Acid and Base Concept PDF – Uses, Examples, Indicators, PH Value
Acid Definition
- These are the substance which have sour taste and turn blue litmus red.
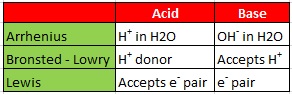
- These are the substance which gives H+ ions in their aqueous solution (Arrenius concept) e.g. HCL or which gives proton (Bronsted Lowry concept) e.g., CH3COOH or which accepts electrons (Lewis concepts) e.g., BF3, AlCl3, NA+, K+, PF3, SF4, PF5 etc.
- Acids give hydrogen with more reactive metals, e.g., Zn (metal) + 2HCl (acid) → ZnCl2 (Salt) + H2 (Hydrogen)
- Acids give carbon dioxide gas (CO2) with carbonates e.g., CaCO3 (marble or calcium carbonate) + H2SO4 (sulphuric acid)→ CaSO4 + H2O + CO2
- These in aqueous solution are conductor of electricity.
Sources of Some Important Acids
Acids have many sources which are important to know for health and industries related.
| Acid | Source |
| Citric acid | Lemon, orange, grapes |
| Lactic acid | Sour milk |
| Butyric acid | Rancid butter |
| Tarteric acid | Grapes |
| Acetic acid | Vinegar |
| Maleic acid | Apples |
| Stearic acid | Fats |
| Oxalic acid | Tomato, wood sorrel |
| Carbonic acid | Soda water aerated drinks |
Uses of Some Acids
Below are given some acid usages.
Hydrochloric acid (HCL)
- It is present in gastric juices and is responsible for the digestion.
- It is used as bathroom cleaner, as pickling agent, tanning of leather, in dying and in the manufacture of gelatine from bones.
Nitric Acids (HNO3)
- It is used for the manufacture of fertilizers like NH4NO3 ; explosive like TNT, picric acid, dynamite etc. rayon, dyes and drugs.
- Also used in the purification of gold and silver.
Sulphuric Acids (H2SO4)
- It is also known as oil of vitriol and is used in manufacture of fertilizers, drugs, detergents and explosive.
Others Uses
- Acetic acid (CH3 COOH) is used in Vineger, medicines and as a solvent.
- Concentrated H2SO4 and HNO3 is used to wash iron for its galvanization.
- Oxalic acid is used to remove rust spot.
- Boric acid is a constituent of eye wash.
- Formic acid is present in red ants.
- Uric acid is present in urine of mammals.
Strength of Acids
There are two types of acids on the basis of strength.
Strong Acids
- Completely ionised in water. Examples – HCL, HNO3, H2SO4
Weak Acids
- Partially ionised in water. Examples – CH3COOH, H2CO3, HCOOH
Classification of Acids
Hydra acids : NH3, H2S, HCl, HBr, HF
Oxy acids : HNO3, H2SO4, HCL4O, HIO4
Basicity of an acid
- The number of removable hydrogen ions from an acid is basicity of that acid.
- Mono basic acid (one removable H+ ion) → HCl, HNO3
- Dibasic acid (two removable H+ ion) →
- Tribasic acid (three removable H+ ion) →
- Acid strength (1. HF < HCL < HBr < HI) or (2. CH3COOH < H2SO4 < HNO3 < HCl)
Base Definition
- These are the substances which have bitter taste and turn red litmus blue.
- Bases give OH- ion in their aqueous solution (Arrhenius concept) e.g., NaOH, KOH, CsOH; Mg(OH)2 etc., or accept proton (Bronsted-Lowry concept) e.g., NH3, H2O etc., or donate electrons (Lewis concept) e.g., simple anions like Cl-, F-, OH-, molecules with unshared (Ione) pairs of electrons like NH3, ROH, R2O, pyridine etc.
- Oxides & hydroxides of metals are bases.
- Water soluble bases are called alkali e.g. NaOH, KOH, etc.
- All alkalies are bases but all bases are not alkalies because all bases are not soluble in water.
Strength of Bases
Strong Bases – NaOH, KOH
Weak Bases – NH4OH, Fe(OH)3
Acidity of a base
The number of removable hydroxyl (OH–) ions from a base is called acidity of a base.
Acidity of NaOH = 1
Acidity of KOH = 1
Acidity of Ca(OH)2 = 2
Indicators
- These are the substances which gives different colors in acid and base solutions.
- Some indicators and their colour in acid and base medium are
| Indicator | Colour | |
| In Acid | In Base | |
| Phenolpthalein | Colourless | Pink |
| Methyl orange | Red | Yellow |
| Methyl red | Red | Yellow |
| Phenol red | Yellow | Red |
- Water soluble bases are called alkali e.g., NaOH, KOH.
- Basicity is the number of replacable OH- ions. e.g., it is 1 for NaOH, 2 for Ca(OH)2.
PH Value
- It is a measure of acidity or basicity of a solution.
- It is defined as the negative logaritham of the concentration in (mol/L) of hydrogen ions which it contains, i.e.,
pH = – log[H+] = log (1/[H+]).
or [H+] = 1 × 10-pH
- It is 7 for neutral solution, greater than 7 for basic solution and less than 7 for acidic solution.
- pH of some common substances are –
| Substance | pH |
| Gastric juice | 1.0 – 3.0 |
| Soft drink | 2.0 – 4.0 |
| Lemon | 2.2 – 2.4 |
| Vinegar | 2.4 – 3.4 |
| Urine (human) | 4.8 – 8.4 |
| Saliva (human) | 6.5 – 7.5 |
| Rain water | 6.0 |
| Tears | 7.4 |
| Sea water | 8.5 |
| Milk of magnesia | 10.5 |
| Blood plasma (human) | 7.30 – 7.42 |
| Milk | 6.5 |
| Wine | 2.5 |
Importance of pH in everyday life
- Our body works within a narrow pH range of 7.0 to 7.8. Plants and animal also survive in a narrow range.
- In digestive system : Hydrochloric acid is produced in the stomach which helps in the digestion of food but if it becomes excess, the pH falls, and pain and irritation occurs. To get rid of this ANTACIDS like milk of magnesia (Mg(OH)2) is generally used to adjust the pH.
- In saving tooth decay : Substances like chocolates and sweets are degraded by bacteria present in our mouth. When the pH falls to 5.5, tooth decay starts. Tooth enamel (calcium phosphate) is the hardest substances in our body and it gets corroded. The saliva produced by salivary glands is slightly alkaline, it helps to increase the PH, to some extent, but tooth paste is used to neutralise excess acid in the mouth.
Acid and Base Concept with Example PDF
Practice make a man perfect. All the best for your upcoming exam!
You can join or visit at ![]() or
or ![]() or
or ![]() for always keep in touch with further updates.
for always keep in touch with further updates.
